







《《氮及其化合物》化工生产中的重要非金属元素(第2课时氨和铵盐)PPT课件下载》是由用户上传到老师板报网,本为文库资料,大小为1.13 MB,总共有50页,格式为pptx。授权方式为VIP用户下载,成为老师板报网VIP用户马上下载此课件。文件完整,下载后可编辑修改。
- 文库资料
- 50页
- 1.13 MB
- VIP模板
- pptx
- 数字产品不支持退货
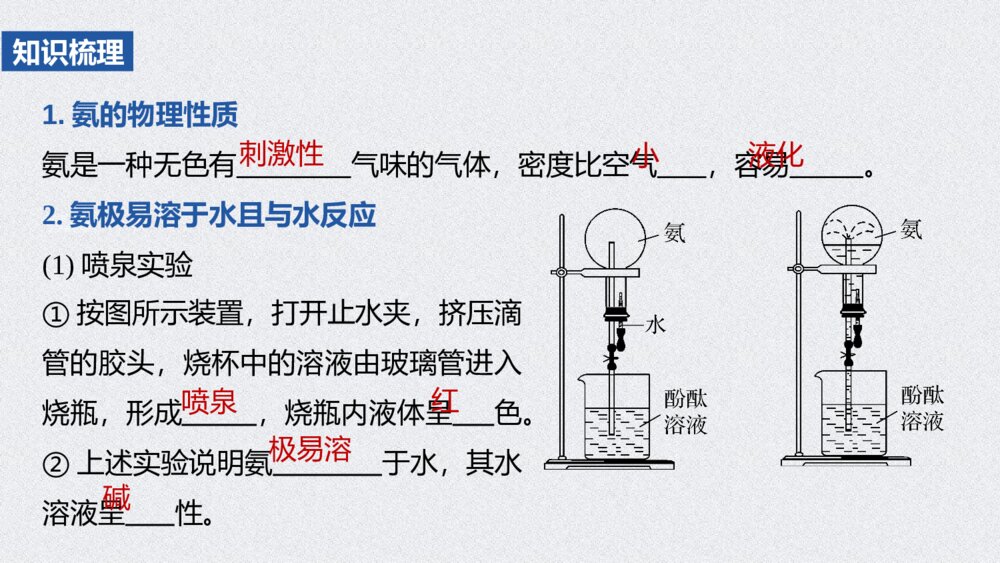
第2课时 氨和铵盐第五章 第二节 氮及其化合物1.能从物质类别、氮元素价态的角度,认识氨、铵盐的性质与转化,促进“证据推理与模型认知”化学核心素养的发展。2.设计实验,如氨的性质实验、制备实验,铵盐的性质实验及铵离子的检验等,实现氨的转化与生成,增强“科学探究”意识。核心素养发展目标二、铵盐的性质与检验内容索引NEIRONGSUOYIN一、氨的性质三、氨的实验室制法氨的性质ANDEXINGZHI01PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/1.氨的物理性质氨是一种无色有气味的气体,密度比空气,容易。2.氨极易溶于水且与水反应(1)喷泉实验知识梳理①按图所示装置,打开止水夹,挤压滴管的胶头,烧杯中的溶液由玻璃管进入烧瓶,形成,烧瓶内液体呈色。②上述实验说明氨于水,其水溶液呈性。刺激性小液化喷泉红极易溶碱PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(2)氨与水的反应①氨气易与水反应:,其水溶液称为氨水。在氨水中:,氨水具有性,能使石蕊溶液变蓝。②氨水中的氮元素主要以的形式存在,只有少量的NH3·H2O电离为NH和OH-,但在求氨水的物质的量浓度、质量分数时应将视为溶质。③NH3·H2O不稳定,受热易分解:,因此,氨水应密封保存在阴凉处。NH3+H2ONH3·H2ONH3·H2ONH+4+OH-弱碱NH3·H2O+4NH3NH3·H2O=====△NH3↑+H2ONH3+H2ONH3·H2ONH3·H2ONH+4+OH-+4NH3·H2O=====△NH3↑+H2OPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/3.氨与酸反应(1)氨与氯化氢相遇时的现象是,反应的化学方程式是。(2)工业上用氨、二氧化碳和水制取碳酸氢铵的化学方程式是。(3)工业上用硫酸吸收氨的化学方程式是。产生白烟NH3+HCl===NH4ClNH3+CO2+H2O===NH4HCO32NH3+H2SO4===(NH4)2SO4PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/4.氨具有还原性(1)请写出氨气与氧气在催化剂作用下生成NO的化学方程式:。(2)氨气可被氧化铜氧化为氮气,反应的化学方程式为。4NH3+5O2======催化剂△4NO+6H2O2NH3+3CuO=====△N2+3Cu+3H2O======催化剂△=====△PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(1)液氨可用作制冷剂,是因为其汽化时吸收大量的热( )(2)氨水能导电,所以氨水是电解质( )(3)新制饱和氨水中含氮粒子物质的量浓度最大的是NH( )(4)将蘸有浓氨水的玻璃棒靠近浓硫酸有白烟产生( )(5)氨中氮元素的化合价为-3价,在反应中只能升高而具有还原性( )判断正误√×××√+4+4PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/思维启迪液氨、氨水、一水合氨的区别名称液氨氨水一水合氨物质类别纯净物氢化物非电解质混合物氨的水溶液溶质为氨纯净物一元弱碱电解质粒子种类NH3NH3·H2O、NH3、H2O、、OH-、H+NH3·H2ONH+4NH+4PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/主要性质不导电不具有碱性能导电具有碱性极易分解具有碱性存在条件常温常压下不存在常温常压下可存在常温常压下不存在PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/1.喷泉是一种常见的自然现象,其产生的原因是存在压强差。深度思考装置中的气体a可能为①氯气、②氢气、③氨气、④氯化氢、⑤二氧化碳。打开装置中的止水夹,挤压胶头滴管,若液体b为水时,能产生喷泉现象的气体a可能是(填序号,下同);若液体b为氢氧化钠浓溶液时,能产生喷泉现象的气体a可能是。③④①④⑤PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/归纳总结产生喷泉的原理分析产生喷泉的原因为气体a易溶于液体b或易和b反应,造成烧瓶内气体物质的量减小,压强急剧减小。PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/2.若将22.4L氨气(标准状况)溶于水,得到500mL氨水。(1)氨溶于水反应的化学方程式为。氨水呈碱性的原因是。(2)该氨水中的含氮粒子有,它们的物质的量浓度之和为。(3)写出氨水与下列物质反应的离子方程式:①稀硫酸:;②氯化铁溶液:。NH3+H2ONH3·H2ONH3·H2O部分电离产生OH-NH3、NH3·H2O、NH+42mol·L-1NH3·H2O+H+===NH+4+H2O3NH3·H2O+Fe3+===Fe(OH)3↓+3NH+4NH3+H2ONH3·H2ONH3、NH3·H2O、NH+4NH3·H2O+H+===NH+4+H2O3NH3·H2O+Fe3+===Fe(OH)3↓+3NH+4PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/3.工业上常用氮气与氢气合成氨,再以氨为原料制取硝酸,请写出有关反应的化学方程式,并说明各反应中含氮物质发生的是氧化反应还是还原反应。2NO+O2===2NO2 NO发生氧化反应3NO2+H2O===2HNO3+NO NO2既发生氧化反应又发生还原反应提示N2+3H2高温、高压催化剂2NH3N2发生还原反应4NH3+5O2=====催化剂△4NO+6H2ONH3发生氧化反应提示N2+3H2高温、高压催化剂2NH3N2发生还原反应4NH3+5O2=====催化剂△4NO+6H2ONH3发生氧化反应PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/相关链接喷泉实验及装置(1)喷泉实验原理喷泉产生的本质原因是烧瓶内外形成压强差,由于烧瓶内气体的压强小于烧瓶外的压强,所以液体会被压入烧瓶内形成喷泉。产生气压差的方法有:①减小烧瓶内气压,如液体将气体吸收或与其反应等;②增大烧瓶外压强。PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(2)常见装置图①图甲装置形成“喷泉”是因为烧瓶内气体极易溶于烧杯和胶头滴管中的液体,使烧瓶内的压强减小而产生压强差,烧杯中的液体被压入烧瓶内形成“喷泉”。PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/②图乙装置形成“喷泉”可采用使烧瓶受热的方法,瓶内气体膨胀,打开止水夹,止水夹下部导管中的空气受热排出,烧瓶内的气体与液体接触而溶解,使烧瓶内压强减小形成压强差而形成“喷泉”。③图丙装置中锥形瓶内的物质反应产生气体(或锥形瓶中液体受热挥发出气体),使锥形瓶内气体压强增大而产生压强差,将液体从锥形瓶中压入烧瓶中形成“喷泉”。解析 氨气易液化,液氨常用来作制冷剂;氨气密度比空气小(常用向下排空气法收集);氨气极易溶于水(可用来做喷泉实验);氨气溶于水,能与水反应生成电解质一水合氨,其水溶液能导电,但氨气是非电解质。1.关于氨的下列叙述中,错误的是A.氨易液化,液氨常用来作制冷剂B.氨气易溶于水,可用来做喷泉实验C.实验室常用向下排空气法收集氨气D.氨气溶于水能导电,氨气是电解质跟踪强化123√2.氨水的碱性较弱,其原因是A.它能使酚酞溶液变红色B.氨水极易挥发出氨C.氨与水结合生成的一水合氨(NH3·H2O)只有少部分电离成NH和OH-D.溶于水的氨大部分以NH3分子形式存在解析 NH3溶于水后,大部分结合成一水合氨的形式,只有少部分NH3·H2O电离成NH和OH-,因此溶液碱性较弱,C项正确。123+4√+4+4+43.下列关于氨气、液氨、氨水的叙述正确的是A.氨气、液氨、氨水的成分相同,均为纯净物B.蘸有浓盐酸的玻璃棒与蘸有浓氨水的玻璃棒相互靠近,有白烟生成C.氨水的密度比水小,且浓度越大,密度越大D.氨水的溶质为NH3·H2O,是氨水中含量最多的微粒123√解析 氨水是混合物,氨水的密度随浓度的增大而减小,氨水中水的含量最多。返回02铵盐的性质与检验ANYANDEXINGZHIYUJIANYANPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/1.铵盐及其性质(1)常见的铵盐①与形成的化合物称为铵盐。②农业上常用的铵态氮肥,如等都属于铵盐,它们都是溶于水的晶体。(2)铵盐不稳定,受热易分解NH4Cl,NH4HCO3。知识梳理铵根离子(NH+4)酸根离子NH4HCO3、(NH4)2SO4、NH4NO3易=====△NH3↑+HCl↑=====△NH3↑+CO2↑+H2O铵根离子(NH+4)=====△=====△PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(3)铵盐与碱反应2NH4Cl+Ca(OH)2。NH4NO3+NaOH。=====△CaCl2+2NH3↑+2H2O=====△NaNO3+NH3↑+H2O=====△=====△PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/2.铵盐(NH)的检验(1)原理+4+4PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(2)方法①NaOH溶液法取少量固体样品或溶液于试管中,再加入浓的NaOH溶液,加热产生能使湿润的红色石蕊试纸变蓝的气体(或将蘸有浓盐酸的玻璃棒靠近管口,有白烟产生),证明固体样品或溶液中含有NH。②碱石灰法把碱石灰与某物质的固体混合物在研钵里研磨,产生的气体能使湿润的红色石蕊试纸变蓝(或将蘸有浓盐酸的玻璃棒靠近研钵口,有白烟产生),则可以判断该物质是铵盐。+4+4PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/1.向盛有少量硫酸铵溶液(或氯化铵溶液、硝酸铵溶液)的试管中加入氢氧化钠溶液并加热,用镊子夹住一片湿润的红色石蕊试纸放在试管口,观察到的现象是,反应的离子方程式是。深度思考红色石蕊试纸变蓝NH+4+OH-=====△NH3↑+H2ONH+4+OH-=====△NH3↑+PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/2.铵盐溶液与碱溶液反应的离子方程式(1)铵盐溶液与烧碱溶液混合后加热:。(2)铵盐稀溶液与烧碱稀溶液混合:。(3)铵盐浓溶液与烧碱浓溶液混合:。NH+4+OH-=====△NH3↑+H2ONH+4+OH-===NH3·H2ONH+4+OH-===NH3↑+H2ONH+4+OH-=====△NH3↑+H2ONH+4+OH-===NH3·H2ONH+4+OH-===NH3↑+H2OPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/3.根据铵盐的性质,分析应当怎样合理地储存和施用铵态氮肥?提示 贮存铵态氮肥时,为了防止受热分解,应密封包装并放在阴凉通风处;施肥时应将铵态氮肥埋在土下,且不能与碱性物质(如草木灰)混用。PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/特别提示(1)铵盐受热分解都有氨生成,但硝酸铵受热分解最终生成氮气或氮氧化物。(2)分别用试管加热氯化铵和单质碘时,都由固体变为气体,而在试管口遇冷又凝结为固体,其本质不同,前者为化学变化,后者为物理变化。(3)检验溶液中含有NH时,需要加入浓碱溶液并加热,其目的是利用氨气的生成与检验。+4+4PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/相关链接铵盐受热分解的产物(1)如果组成铵盐的酸是易挥发或易分解的酸,则固体铵盐受热分解时,氨气与酸一起挥发,冷却时又重新结合成铵盐。如NH4Cl、NH4HCO3等。(2)如果组成铵盐的酸是难挥发性酸,则固体铵盐受热分解时,只有氨呈气态逸出,而难挥发性酸残留在容器中,如(NH4)2SO4分解的化学方程式为(NH4)2SO4NH3↑+NH4HSO4或(NH4)2SO42NH3↑+H2SO4。=====△=====△=====△=====△PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(3)如果组成铵盐的酸是具有强氧化性的酸,则在较低的温度下慢慢分解可得到NH3和相应的酸,如NH4NO3。由于生成的氨气具有还原性,硝酸具有氧化性,生成的NH3易被HNO3氧化,则反应时的温度不同,形成氮的化合物也不同,如将NH4NO3在不同的温度下加热分解分别可得到N2O、NO2、N2O3、N2等。1.下列关于铵盐的说法中正确的是A.所有铵盐受热均可以分解,其产物中都有氨B.所有铵盐都易溶于水,所有铵盐中的N均呈-3价C.氯化铵溶液中加入烧碱浓溶液共热时反应的离子方程式为NH+OH-NH3↑+H2OD.氯化铵和氯化钠的固体混合物可用升华法分离跟踪强化123+4=====△√+4=====△解析 A项,有的铵盐受热分解产生氨(如NH4Cl),有的铵盐受热分解最终不产生氨(如硝酸铵);D项,可用加热法分离NH4Cl和某些固体(不分解、不升华的固体)的混合物,但其过程为先受热分解,再降温化合,而不是升华。1231232.下列离子方程式书写正确的是A.氯化铵固体和熟石灰混合加热:NH+4+OH-=====△NH3↑+H2OB.NaOH溶液与NH4Cl溶液混合加热:NH+4+OH-=====△NH3·H2OC.氨水中加入盐酸:NH3·H2O+H+===NH+4+H2OD.氯化铝溶液中加入过量的氨水:Al3++3OH-===Al(OH)3↓√2.下列离子方程式书写正确的是A.氯化铵固体和熟石灰混合加热:NH+4+OH-=====△NH3↑+H2OB.NaOH溶液与NH4Cl溶液混合加热:NH+4+OH-=====△NH3·H2OC.氨水中加入盐酸:NH3·H2O+H+===NH+4+H2OD.氯化铝溶液中加入过量的氨水:Al3++3OH-===Al(OH)3↓123B项,加热条件下NH3·H2O应分解为NH3和H2O,离子方程式应为NH+4+OH-=====△NH3↑+H2O,错误;解析 A项,该反应属于固体间的加热反应,不能写成离子形式,错误;C项,符合反应原理及离子方程式书写要求,正确;D项,一水合氨是弱碱,不能完全电离,向氨水中滴加氯化铝溶液应生成Al(OH)3沉淀,离子方程式应为Al3++3NH3·H2O===Al(OH)3↓+3NH+4,错误。B项,加热条件下NH3·H2O应分解为NH3和H2O,离子方程式应为NH+4+OH-=====△NH3↑+H2O,错误;D项,一水合氨是弱碱,不能完全电离,向氨水中滴加氯化铝溶液应生成Al(OH)3沉淀,离子方程式应为Al3++3NH3·H2O===Al(OH)3↓+3NH+4,错误。3.(2019·伊春市二中期末)检验铵盐的一般方法是将待测物取少许置于试管中,然后A.加水溶解后,再滴入酚酞溶液B.加强碱溶液加热后,用湿润的红色石蕊试纸在试管口检验C.加入强酸溶液加热后,再滴入酚酞溶液D.加入强碱溶液加热后,再滴入酚酞溶液123解析 将固体放入试管中,加强碱溶液加热后,用湿润的红色石蕊试纸放在试管口,湿润的红色石蕊试纸会变蓝色,证明产生的气体是氨,则该盐中含有铵根离子,B正确。返回√03氨的实验室制法ANDESHIYANSHIZHIFAPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/实验室制取氨的实验装置如图所示:知识梳理(1)反应原理:。(2)碱石灰的作用:。(3)由于氨极易溶于水,不能用排水法收集,常用法来收集。2NH4Cl+Ca(OH)2=====△CaCl2+2NH3↑+2H2O吸收水蒸气,干燥氨气向下排空气2NH4Cl+Ca(OH)2=====△CaCl2+2NH3↑+2H2OPPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/(4)验满方法①把湿润的红色石蕊试纸置于试管口,试纸变;②将蘸有浓盐酸的玻璃棒置于试管口,有产生。(5)尾气处理可在导管口放一团用浸湿的棉花球。蓝色白烟水或稀硫酸PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/1.制取氨气的发生装置与实验室制取哪种气体的相同?使用该装置需注意什么问题?提示 与实验室制取氧气的发生装置相同。该套装置在使用时要特别注意加热时要使试管口略低于管底,以防止冷凝水倒流使试管炸裂。2.在制取氨的实验装置中,使用棉花的作用是什么?提示 若为干燥的棉花则起到减缓氨气与空气对流的作用;若为蘸有水或稀硫酸的棉花,则还可以吸收多余的NH3。深度思考PPT模板:www.1ppt.com/moban/PPT素材:www.1ppt.com/sucai/PPT背景:www.1ppt.com/beijing/PPT图表:www.1ppt.com/tubiao/PPT下载:www.1ppt.com/xiazai/PPT教程:www.1ppt.com/powerpoint/资料下载:www.1ppt.com/ziliao/个人简历:www.1ppt.com/jianli/试卷下载:www.1ppt.com/shiti/教案下载:www.1ppt.com/jiaoan/手抄报:www.1ppt.com/shouchaobao/PPT课件:www.1ppt.com/kejian/语文课件:www.1ppt.com/kejian/yuwen/数学课件:www.1ppt.com/kejian/shuxue/英语课件:www.1ppt.com/kejian/yingyu/美术课件:www.1ppt.com/kejian/meishu/科学课件:www.1ppt.com/kejian/kexue/物理课件:www.1ppt.com/kejian/wuli/化学课件:www.1ppt.com/kejian/huaxue/生物课件:www.1ppt.com/kejian/shengwu/地理课件:www.1ppt.com/kejian/dili/历史课件:www.1ppt.com/kejian/lishi/3.氨气是化学实验室常需制取的气体。实验室制取氨气通常有两种方法:①用固体氢氧化钙与氯化铵共热反应;②在常温下用固体氢氧化钠与浓氨水混合反应。(1)下面的发生装置图中,方法①应选用装置(填“A”或“B”,下同),方法②应选用装置。AB解析 装置A适用于固固混合加热制取气体,装置B适用于固液混合不加热制取气体,故方法①选A装置,方法②选B装置。(2)在制取氨气后,如果要干燥氨气,应选用的干燥剂是(填字母)。A.浓硫酸B.固体氢氧化钠C.五氧化二磷D.稀硫酸B解析 氨气是碱性气体,可与硫酸、P2O5反应。(3)如图是收集氨气的几种装置,其中可行的是,集气的原理是________。D解析 氨气极易溶于水,且密度小于空气,故采用向下排空气法收集,其中B装置排不出空气。利用氨气的密度小于空气,采用短管进氨气,长管出空气,即可收集氨气特别提示(1)制取氨时,不能用硝酸铵代替氯化铵。因为硝酸铵在加热过程中可能发生爆炸性的反应,发生危险。(2)制取氨时,不能用氢氧化钠代替熟石灰。因为氢氧化钠具有吸湿性,易结块,不利于产生NH3,且在加热条件下易腐蚀试管。(3)氨气是碱性气体,不能用酸性干燥剂(浓硫酸等)干燥,也不能用无水CaCl2干燥,因为它们均能与氨气发生反应,常用碱石灰作干燥剂。相关链接快速制取氨气的方法方法化学方程式(或原理)气体发生装置加热浓氨水NH3·H2OH2O+NH3↑=====△=====△浓氨水+固体NaOHNaOH溶于水放热,促使氨水分解,且OH-浓度的增大有利于NH3的放出浓氨水+固体CaONH3·H2O+CaO===NH3↑+Ca(OH)2CaO的作用:①吸水后放热促进NH3的放出;②增加溶液中OH-的浓度,减小NH3的溶解度1.(2019·贵阳高一检测)在实验室里,某学习小组设计了下列制取纯净干燥氨气的方案,简易、可行的方案是A.加热氯化铵固体,再通过碱石灰B.在氮气和氢气的混合气体中加入铁触媒,并加热至500℃,再通过碱石灰C.加热浓氨水,再通过浓硫酸D.在生石灰中加入浓氨水,再通过碱石灰跟踪强化12√2.实验室制取少量干燥的氨气涉及下列装置,其中正确的是A.①是氨气发生装置B.②是氨气吸收装置C.③是氨气发生装置D.④是氨气收集、检验装置√12解析 ①中氯化铵固体受热分解生成氨气和氯化氢,而在试管口温度降低,氨气和氯化氢又重新化合成固体氯化铵,无法得到氨气;②中漏斗没入水下起不到防倒吸的作用,应使倒置漏斗的上边缘刚好没入水中;③氧化钙能与浓氨水中的水反应生成氢氧化钙,同时放出大量热,使浓氨水分解放出氨气;④中的收集装置应在试管口塞一团棉花,防止因空气对流,而使氨气逸出。返回12



















